Therapeutic areas
The ANXV journey towards helping patients begins in the eye
ANXV has the potential of being used broadly in medicine - as a therapeutic agent, as a carrier for other therapeutic agents, and in molecular imaging. The therapeutic potential of ANXV is broad within several indications in different therapeutic areas. However, our first step is to investigate ANXV´s potential effect in the eye disease retinal vein occlusion.
Therapeutic areas and clinical studies
Focus areas for development
ANXV is an investigational new drug, and its mode of action is expected to be beneficial for treating both Retinal Vein Occlusion (RVO) and cancer. ANXV is currently being investigated in a Phase 2 study for RVO, a disease that causes vision impairment or blindness in millions of patients every year.
ANXV and retinal vein occlusion
Retinal Vein Occlusion (RVO) is the blockage or closing of a vein in the eye. It is the second most common sight-threatening retinal vascular disorder after diabetic retinopathy. Globally, the prevalence of RVO is reported to be over 16 million cases. RVO might present with a sudden, painless loss of vision and macular oedema (ME). ANXV in RVO is being investigated in an ongoing Phase 2 study with recently diagnosed RVO patients.
Read more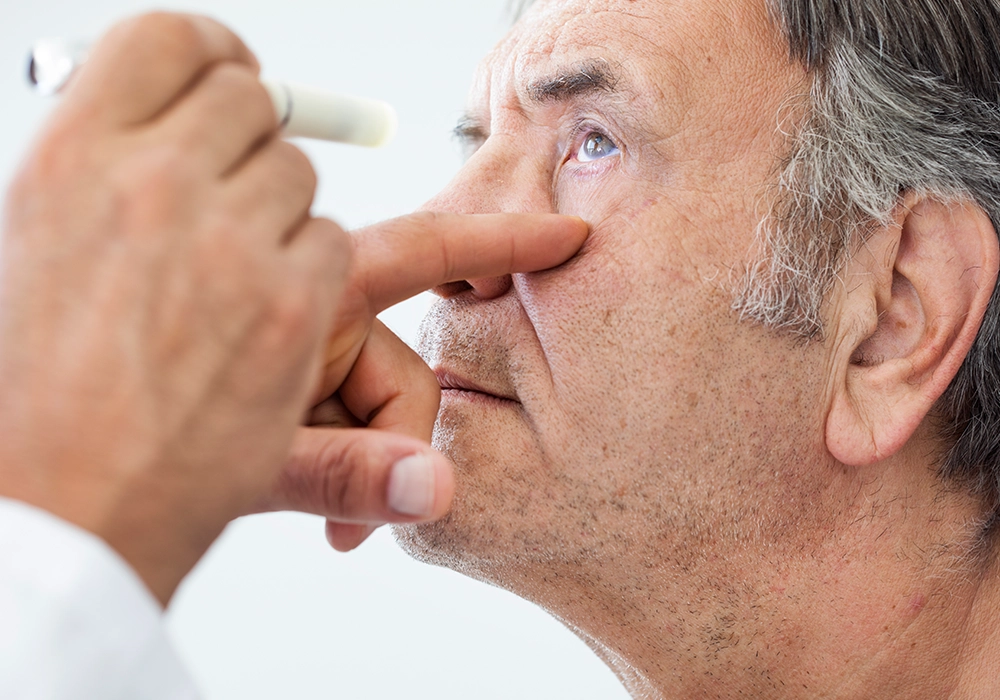
ANXV and cancer
Cancer is a leading cause of death worldwide, accounting for nearly 10 million losses of life in 2020, which accounts for nearly one in six deaths. The most common cancers are those occurring in the breast, lung, colon and rectum, and prostate. There are still significant, and yet unmet, clinical needs, where ANXV could potentially fill a gap. The potential of ANXV in oncology is currently being investigated in the pre-clinical stage.
Read more

ANXV potential in Sickle Cell Disease
Sickle Cell Disease (SCD) is a hereditary disease which affects the red blood cells, resulting in impaired blood supply, damage to organs, severe pain and causes premature death.
SCD patients experience frequent, painful sickle cell crises when sickle cells block blood vessels in different parts of the body. Such crises require intensive care and pain relief. There are currently no treatments that can prevent or stop these crises. However, Annexin A5 has been shown to stop ongoing sickle cell crises in animal models. If ANXV shows an effect in SCD patients, it would fill a major medical need.

ANXV potential in Cardiovascular diseases
Cardiovascular disease is the collective term for diseases that affect the heart and/or the blood vessels. It is the most common cause of death, both in Sweden and in most industrialised countries.
Common examples are myocardial infarction, peripheral occlusive arterial disease (PAOD) and stroke, often caused by atherosclerosis. Atherosclerosis is a chronic inflammatory disease of the blood vessels, and there is still no anti-inflammatory medication available to treat such conditions. However, Annexin A5 has demonstrated desirable effect in preclinical models.

